Good Laboratory Practice (GLP)

GLP Inspections by Monitoring Authorities over the years
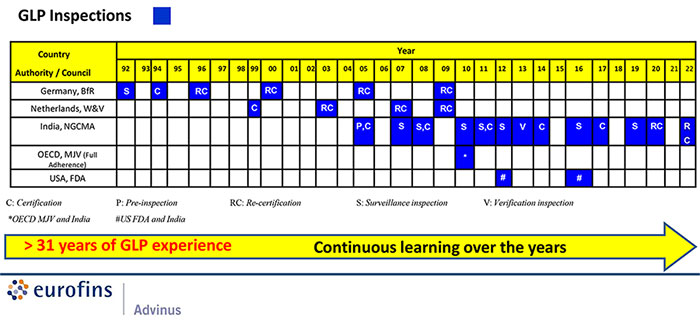
Subsequent to India gaining Full Adherent status to Mutual Acceptance of Data (MAD) in March 2011, GLP inspection for accreditation will only be by the Indian Monitoring Authority.
- GLP accredited since 1992 (Germany, Netherlands and India). The first facility in India to receive GLP accreditation. (Through erstwhile Rallis Research Centre (RRC), which was acquired by Advinus in 2005)
- Since 2011, India is “Full Adherent” to Mutual Acceptance of Data (MAD) as per OECD. Eurofins Advinus was the site of inspection for the OECD Mutual Joint Visit (MJV) with the Indian National GLP Compliance Monitoring Authority (NGCMA). This inspection was instrumental for India’ achieving “Full Adherent” status of MAD.
- Eurofins Advinus adopts OECD Principles of GLP and conducts studies in compliance with OECD GLP. Eurofins Advinus data have been successfully submitted to US FDA, US EPA, EMA, MHRA, DCGI and other global regulatory agencies.
AAALAC International
- AAALAC accredited since 2001; the first facility in India to receive the AAALAC accreditation.
Central Insecticides Board & Registration Committee (CIB&RC)
- Approved for generation of data in Chemistry; Bio-assay including residue studies, persistence and metabolism; Toxicology and Packaging.
Atomic Energy Regulatory Board (AERB)
- Approval for use of radiolabelled compounds for research and development
Committee for the Purpose of Control And Supervision of Experiments on Animals (CPCSEA)
- Approval for research for commercial purpose and breeding for in-house use on small animals and research for commercial purpose on large animals
International Organization for Standardization (ISO)
- The facility is also certified ISO 17025:2017 for Medical Devices, biomaterials and ophthalmic devices testing
ISO 9001:2015 (Quality Management System)
- Certified ISO 9001:2015 for the activities Contract services in Manufacture, Test and Stability study of Pharmaceutical Intermediates/ NCEs/APIs



